Posted: February 14, 2023
Recapping Road Salt Reduction Week!

Did you catch all our content for Road Salt Reduction Week? Don’t worry if you missed some of it, you can find all the daily facts we shared by reading this blog!
The week of January 30th, 2023, Ottawa Riverkeeper ran Road Salt Reduction Week, sharing facts and stories about road salt, and highlighting our community-based monitoring program. Every morning, we shared a new road salt fact to help you understand why its overuse can be harmful to our waterways and what you can do about it.
Here are all the facts we shared:
FACT #1: Salt is harmful to freshwater ecosystems!
We are kicking off Road Salt Reduction Week with the basics! Yes, excessive quantities of road salt (most commonly NaCl) are harmful to freshwater organisms and the ecosystem as a whole. The danger is especially acute for small creeks and streams in urban environments. But why?
The element (literally!) of road salt that is the most dangerous is the Chloride ion (Cl-). Chloride is extremely persistent once it enters an aquatic environment, and stops many organisms from being able to respire, manage water content, or grow. Once Chloride levels reach certain concentrations, it can cause permanent damage to organisms in the environment.

FACT #2: Chloride results from our monitoring are alarming!
Now you know that Chloride harms freshwater ecosystems, but what does the situation look like in our creeks? Unfortunately, we have bad news. Of all the samples we’ve processed, over 80% have exceeded the threshold which indicate harm to the ecosystem!
When Chloride concentrations go above 120mg/L they are considered ‘chronically’ toxic, meaning these levels are dangerous if they persist for a long period of time. Once concentrations cross 640mg/L, conditions reach the ‘acutely’ toxic threshold, where organisms may come to significant harm even through very brief exposure.
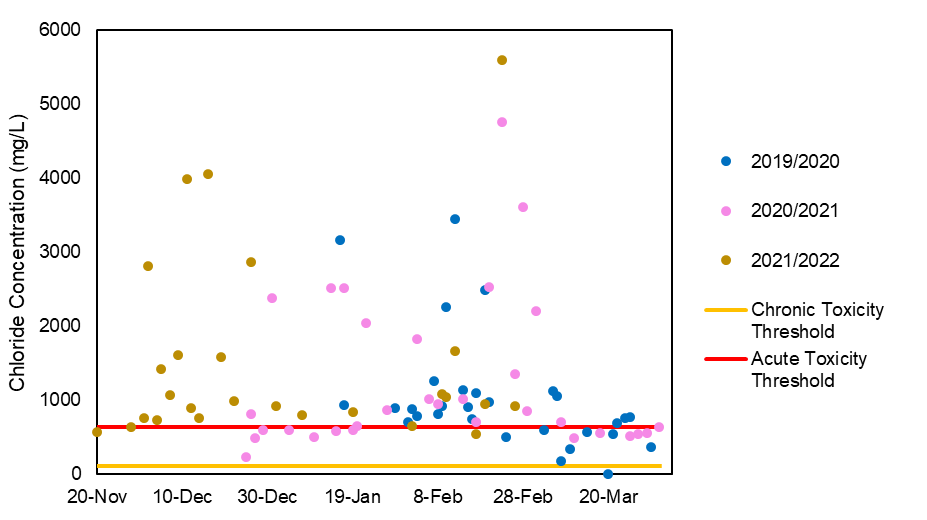
Here is an example from Pinecrest Creek: as you can see, in our three years of monitoring, only one sample was ever below the chronic toxicity threshold, and most were higher than the acute toxicity threshold. Similar results can be found in most of the streams we have monitored.
FACT #3: All Ottawa Riverkeeper data is available to the public!
After sharing some striking numbers from our monitoring initiative, you may be hungry for more. Don’t worry, all of our findings are made publicly available! We want this information to be accessible to anyone who wants to check on their local creek, or to researchers who may want to use this data in their own studies.
All this information (and much more) can be found on Ottawa Riverkeeper’s Open Data Portal, just follow this link!
This data is all collected through community-based monitoring, which means it should belong to the community. We are so grateful to the committed volunteers helping us collect this information. They make this investigation, and all our subsequent work on road salt possible!
FACT #4: Wetlands do not filter out chloride!
February 2nd is World Wetlands Day! Healthy wetlands are a vital part of a healthy watershed. They help to mitigate flooding, keep our rivers clean, and act as crucial habitats for many of our native species.

But how do they stand up to road salt? Unfortunately, although wetlands can act as filters for many pollutants in our waterways, Chloride is not one of them. That means that wetlands are vulnerable to the negative effects of excess road salt use, and don’t act as a shield against this ubiquitous pollutant.
You can help wetlands, and all of our freshwater resources by reducing the amount of salt you use in the winter!
FACT #5: One coffee mug of salt is enough for a two-car driveway!

We have been exploring the negative impacts of road salts and the chloride it introduces to waterways, but what can YOU do about it? Simple, use less!
Many people vastly overuse road salt: since it is a chemical de-icer, you don’t need nearly as much of it as you may think. The one coffee mug rule is a simple way to think about it, but if you want a more technical guideline, then remember that two tablespoons of salt (30 g) is enough for a whole square meter of pavement.
FACT #6: Salt should not be applied for traction!
Road salt is a chemical de-icer; which means it is meant to remove the ice as it dissolves. If you want a better grip on a slippery surface, don’t coat it with grains of salt! You will just be harming the ecosystem. Instead, reach for sand, gravel, or some of the less orthodox options including coffee grounds or cat litter.
What does this look like? Well, for starters your salt should not be piled up. Here is a good rule of thumb: if you are ever stepping on more than seven grains of salt at once, it has been applied too thickly. You want the grains to be spread out evenly so they interact with a larger surface area of ice, without adding excess salt to runoff that enters our waterways.

FACT #7: Salt becomes ineffective as a de-icer at lower temperatures!
Here is our final tip, and it may be one of the most important: don’t use salt when temperatures go below -7 degrees! As this is a common occurrence in our watershed, we really want to hammer this one home.
Why? While in theory, the freezing point of a salt solution is -21 degrees, things don’t work that way in practice. As the pavement gets colder, more salt is required to melt the ice. Somewhere between -7 and -10 degrees celsius, the amount of salt needed becomes so high that it simply becomes an ineffective solution for removing ice.
It is important to note that there are ways around this: for example applying a brine solution can be more effective at lower temperatures (many municipalities use this for their roads). But if all you have is the standard solid rock salt most of us are familiar with, check a thermometer before putting it down!

We would like to thank the Ottawa Community Foundation for providing the funding for our awareness campaign this year.

Thank you for advocating for less salt. It is also incredibly destructive to metal and concrete that make up our buildings, vehicles and infrastructure. We all win when salt is used responsibly.